International Journal of Energy Science and Engineering
Articles Information
International Journal of Energy Science and Engineering, Vol.4, No.2, Jun. 2018, Pub. Date: Aug. 10, 2018
Empirical Study on Effective Groups in Treatment Process of Sour Gas by Novel Method: Introduction of New Producer
Pages: 12-16 Views: 2215 Downloads: 343
[01]
Kasra Amiri, Department of Chemical Engineering, Firoozabad Branch, Islamic Azad University, Firoozabad, Iran.
[02]
Seyyedeh Media Jad Baba, Department of Chemical Engineering, Sirjan Branch, Islamic Azad University, Sirjan, Iran.
[03]
Farshad Farahbod, Department of Petroleum Engineering, Firoozabad Branch, Islamic Azad University, Firoozabad, Iran.
Adsorption of water vapour on the nano zinc oxide bed is a studied method of dehydration, in this work. The amount of mass transfer which varies with the values of temperature, pressure, dimensionless groups in length, 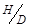 , temperature,
, temperature, 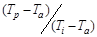 , dimensionless pressure,
, dimensionless pressure, 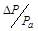 , catalyst diameter, dimensionless diameter,
, catalyst diameter, dimensionless diameter, 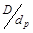 , and initial amount of water in the feed stream is investigated through the experiments. The results are analysed by the historical data tool of Design expert software. The energy optimization is occurred when the process do in optimum conditions. In addition, the experimental data are analysed by historical data interface on the Response Surface tab. This helps to evaluate the experiments and investigate the optimum pressure and temperature. The software suggests linear correlation between variables. The parameters of temperature, pressure,
, and initial amount of water in the feed stream is investigated through the experiments. The results are analysed by the historical data tool of Design expert software. The energy optimization is occurred when the process do in optimum conditions. In addition, the experimental data are analysed by historical data interface on the Response Surface tab. This helps to evaluate the experiments and investigate the optimum pressure and temperature. The software suggests linear correlation between variables. The parameters of temperature, pressure, 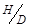 , particle diameter and initial amount of water are chosen as numeric factors. The response is defined as amount of dehumidification flux,
, particle diameter and initial amount of water are chosen as numeric factors. The response is defined as amount of dehumidification flux, 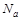 .
.
Energy Optimization, Bed, Dimensionless, Adsorption, Pressure, Temperature
[01]
Prajapat Ganshyam, Rellegadla Sandeep, Jain Shikha, Agrawal Akhil, Reservoir souring control using benzalkonium chloride and nitrate in bioreactors simulating oil fields of western India, International Biodeterioration & Biodegradation, Volume 132, August 2018, Pages 30-39.
[02]
Santosa, Sri Juari, Kunarti, Eko, Karmanto, Sri, 2013. Synthesis and utilization of Mg/Al hydrotalcite for removing dissolved humic acid, Applied Surface Science 254, 7612–7617.
[03]
W. P. Cheng, Comparison of hydrolysis/coagulation behaviour of polymeric and monomeric iron coagulants in humic acid solution, Chemosphere 47 (2013) 963–969.
[04]
Bian Xiao-Qiang, Zhang Lu, Du Zhi-Min, Chen Jing, Zhang Jian-Ye, Prediction of sulfur solubility in supercritical sour gases using grey wolf optimizer-based support vector machine, Journal of Molecular Liquids, Volume 261, 1 July 2018, Pages 431-438.
[05]
Abrahamson, A. B. I., Brunstrom, B., Sundt, R. C., Jorgensen, E. H., 2008. Monitoring contaminants from oil production at sea by measuring gill EROD activity in Atlantic cod (Gadus morhua). Environ. Pollut. 153, 169e175.
[06]
Esmaeilzadeh, F., Goodarznia, I., 2005. Supercritical extraction of phenanthrene in the crossover region, J. Chemical Engineering Data 50, 49–51.
[07]
Beyer, J., Jonsson, G., Porte, C., Krahn, M. M., Ariese, F., 2010. Analytical methods for determining metabolites of polycyclic aromatic hydrocarbon (PAH) pollutants in fish bile: a review. Environ. Toxicol. Pharmacol. 30, 224e244.
[08]
sima Kamari, Hidaka Kohei, Miyanaga Kazuhiko, Tanji Yasunori, The presence of nitrate- and sulfate-reducing bacteria contributes to ineffectiveness souring control by nitrate injection, International Biodeterioration & Biodegradation, Volume 129, April 2018, Pages 81-88.
[09]
Xu. Zhang, Cheng-Fang Zhang, Yi Lin, Kinetics of absorption of CO2 into aqueous solution of MDEA blended with DEA, Ind. Eng. Chem. Res. 2002, 41, 1135-1141.
[10]
Habibi R., Towfighi Daryan J., Rashidi A. M., 2009, Shape and size-controlled fabrication of ZnO nanostructures using noveltemplates, J. Exp. Nanosci. 4 (1) 35-45.
[11]
Alkatheri M., Betancourt-Torcat A., Almansoori A., Comparison of life cycle greenhouse gas emissions from unconventional ultra-sour and conventional gas feedstock for power: A case study of the United Arab Emirates, Journal of Cleaner Production, In press, accepted manuscript, Available online 27 June 2018.
[12]
Habibi R., Rashidi A. M., Towfighi Daryan J., Alizadeh A., 2010, "study of the rod –like and spherical nano ZnO morphology on H2S removal from natural gas". Appl. Surf. Sci., 257, pp. 434-439.
[13]
Tsesmetzis Nicolas, Alsop Eric B., Vigneron Adrien, Marcelis Fons, Lomans Bart P., Microbial community analysis of three hydrocarbon reservoir cores provides valuable insights for the assessment of reservoir souring potential, International Biodeterioration & Biodegradation, Volume 126, January 2018, Pages 177-188.
[14]
Balouria Vishal, Kumar Arvind, Samanta S., Singh A., Debnath A. K., Mahajan Aman, Bedi R. K., Aswal D. K., Gupta S. K., 2013, ‘’Nano-crystalline Fe2O3 thin films for ppm level detection of H2S,’’Sensors Actuators B: Chemical, 181, pp. 471-478.
[15]
Xomeritakis. G, Tasi. C. Y, Brinker. C. J, “Microporous sol-gel derived aminosolicate membrane for enhanced carbon dioxide separation”, Separation and Purification Technology 42, 2005.
[16]
Nejat Tahereh, Movasati Azam, Wood David A., Ghanbarabadi Hassan, Simulated exergy and energy performance comparison of physical–chemical and chemical solvents in a sour gas treatment plant, Chemical Engineering Research and Design, Volume 133, May 2018, Pages 40-54.
[17]
Mandal. B. P, Biswas. A. K., Bandyopadhyay. S. S, “Selective absorption of H2S from gas strems containing H2S and CO2 into aqueous solutions of N-methyldiethanolamine and 2- amino-2- methyl-1- propanol”, Separation and Purification Technology 35, 2004.
[18]
Han Yehua, Zhang Yanfen, Xu Chunming, Hsu Chang Samuel, Molecular characterization of sulfur-containing compounds in petroleum, Fuel, Volume 221, 1 June 2018, Pages 144-158.

ISSN Print: 2381-7267
ISSN Online: 2381-7275
Current Issue:
Vol. 5, Issue 1, March Submit a Manuscript Join Editorial Board Join Reviewer Team
ISSN Online: 2381-7275
Current Issue:
Vol. 5, Issue 1, March Submit a Manuscript Join Editorial Board Join Reviewer Team
| About This Journal |
| All Issues |
| Open Access |
| Indexing |
| Payment Information |
| Author Guidelines |
| Review Process |
| Publication Ethics |
| Editorial Board |
| Peer Reviewers |


